 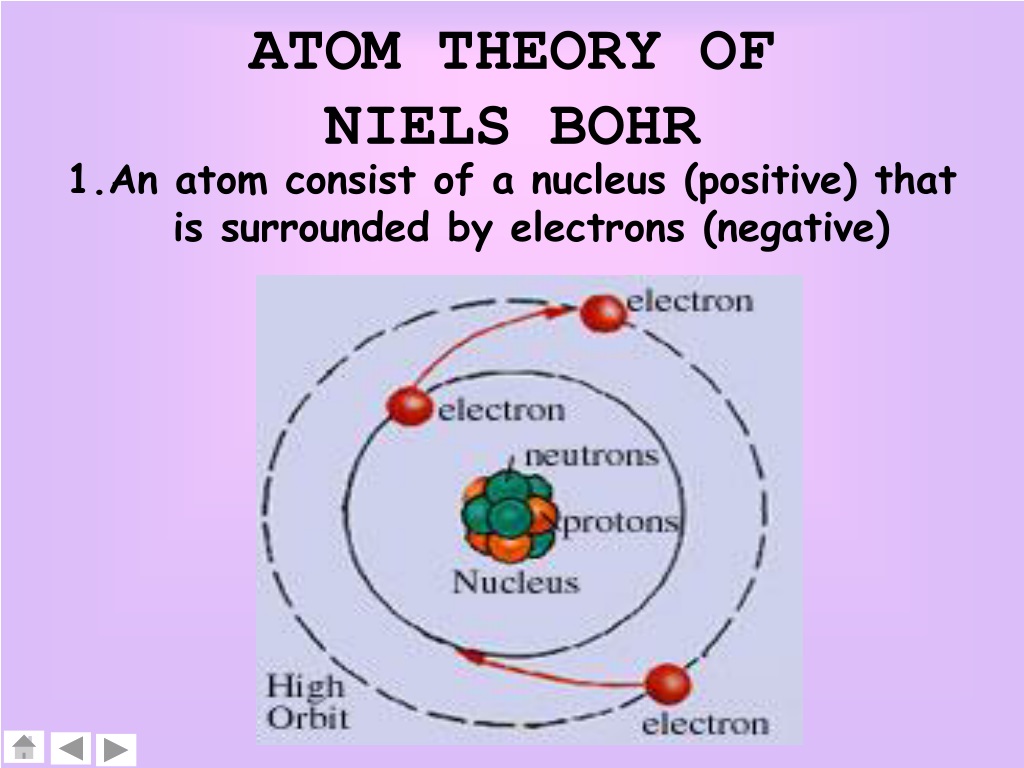
This observation is connected to the discrete nature of the allowed energies of a quantum mechanical system. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. 
It is observed that line spectra discussed in the previous sections show that hydrogen atoms absorb and emit light at only discrete wavelengths. In 1913, a Danish physicist, Niels Bohr (18851962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. From this, it is easy to see that like charges (charges of the same sign) repel each other. The inset shows an electron jumping from orbit n 3 to orbit n 2, emitting a photon of red light with an energy of 1. As Bohr had noticed, the radius of the n 1 orbit is approximately the same size as an atom. Electrons can jump from one orbit to another by emitting or absorbing energy. In Bohr’s model, radius an of the orbit n is given by the formula an h2n2 0 / 2, where 0 is the electric constant. The orbits are labeled by an integer, the quantum number n. It does introduce several important features of all models used to describe the distribution of electrons in an atom. The Bohr model of the atom, a radical departure from earlier, classical descriptions, was the first that incorporated quantum theory and was the predecessor of wholly quantum-mechanical models. Around 400 BC, a Greek philosopher named Democri. Bohr model, description of the structure of atoms, especially that of hydrogen, proposed (1913) by the Danish physicist Niels Bohr. 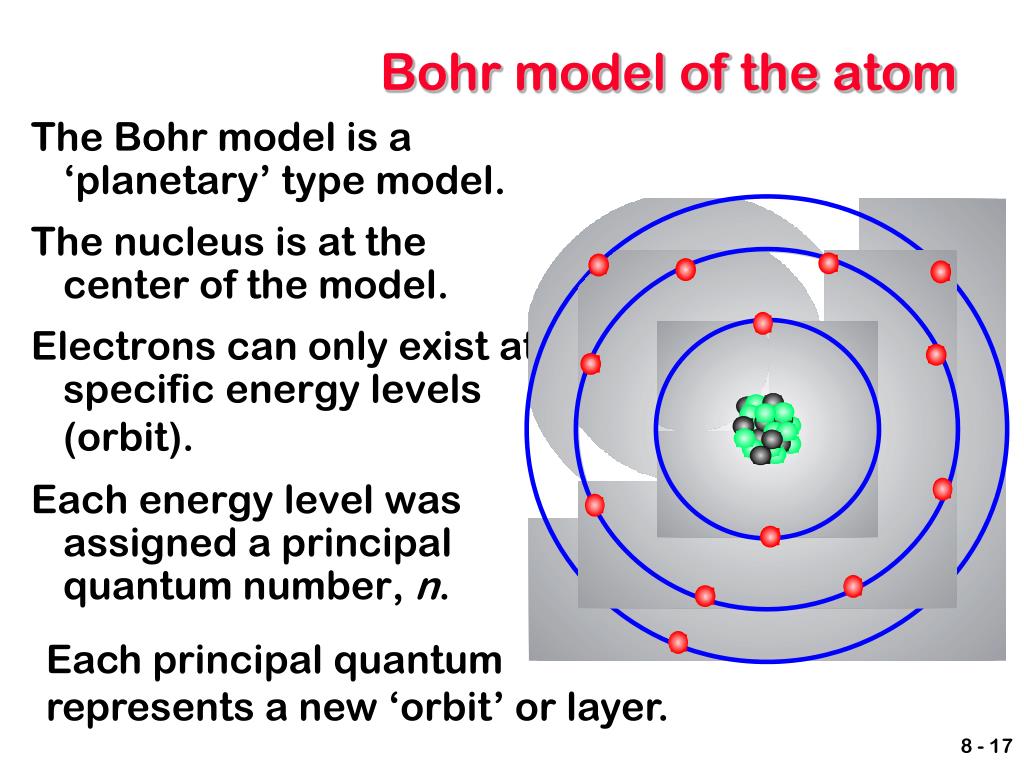
If \(Q_1\) and \(Q_2\) are the same sign, then the curve which is a purely repulsive potential, i.e., the energy increases monotonically as the charges are brought together and decreases monotonically as they are separated. In the Bohr model of the atom, electrons travel in defined circular orbits around the nucleus. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it is does not account for electronelectron interactions in atoms with more than one electron. Atomic Models: Centuries ago, people didn’t know exactly what was inside an atom, but they had some ideas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
